by Anna Lau, PhD, Medical Writer
We generally think about cancer as a disease of individuals, but doing so could limit our view of cancer biology and prevents us from appreciating the complexity of this disease. Cancer can also be a disease of families. There are numerous heritable genetic mutations that greatly predispose families to the development of certain cancers (see here, here, and here).
What if cancer were a disease of a species, a contagious disease that could be passed from person to person through contact? Thankfully, human cancer is not usually contagious—but such cancer is not unheard of. There have been cases of direct transmission of cancer between people (passed between individuals through physical contact): from mother to fetus, from fetus to fetus between twins, and from organ or stem cell donor to recipient via the donated tissue. Let it be clearly emphasized that these are rare events. Thus far, no human cancer has developed the capacity to be easily transmitted through direct contact.
| There are certain cancers that can be spread from person to person through an infectious agent, like a virus. The human papillomavirus can be transmitted through sexual contact and cause many kinds of cancers in both men and women. But this does not mean that the cancer itself is the contagion, only the virus. |
But there are examples in the animal kingdom of contagious cancers that are directly transmissible. One is extraordinarily old, the other quite new, and appreciating their differences and similarities to human cancers can broaden our total understanding of cancer biology.
Devils die, but dogs defy a dreaded disease.
Around 1996, researchers discovered a cancer decimating the Tasmanian devil population. The cancer, termed Tasmanian devil facial tumor disease (DFTD), arose from a single clonal cell lineage in a single devil, but the cancer has spread like wildfire. Symptoms start as lesions on the face, neck, and mouth of affected animals, then enlarge (>3 cm diameter) and ulcerate. Death ensues within months of initial appearance of lesions. Since the emergence of DFTD, the devil population has been devastated, putting the animals at risk of local extinction within just a few decades! Transmission of DFTD occurs through direct contact between animals, during which tumors are transmitted as allografts that are genetically distinct from their hosts. In other words, the cancer is physically passed from one devil to another through normal contact, the cancers examined from different devils are genetically identical (ie, clonal), and the cancers are able to thrive—that is, they do not provoke a host immune response.
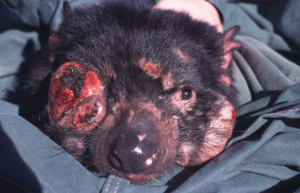
Tasmanian devil with facial tumor disease. Photo by: Menna Jones (creativecommons.org/licenses/by/2.5), via Wikimedia Commons.
There is another clonally transmissible cancer that affects dogs, called canine transmissible venereal tumor (CTVT). CTVT is highly prevalent among dogs living in tropical and subtropical urban environments. Genome sequencing studies have confirmed the clonality of two CTVT tumors from dogs on distant continents and showed this cancer to be the oldest known somatic cell lineage. According to this research, CTVT arose in an ancient wolf or dog about 11,000 years ago and has been passed from dog to dog as allografts ever since then. (One can only imagine what a long, strange trip that has been.) Unlike DFTD, however, CTVT is rarely fatal. Once transferred to a new host, CTVT undergoes an initial progressive phase, marked by rapid tumor growth generally lasting for weeks, then enters a stable phase of slower tumor growth lasting from weeks to indefinitely, and finally settles into a regression phase in most dogs, during which tumors shrink and disappear. In other words, the canine immune system can overcome CTVT.
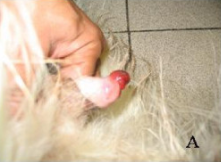
Dog genitals with transmissible venereal tumor. Spugnini EP, et al. J Exp Clin Cancer Res. 2008;27:58.
DFTD and CTVT are strange, but they share properties with human cancers.
For DFTD and CTVT to propagate, they must (a) have a route of transmission, (b) survive and thrive in a new environment, and (c) evade the host immune system in the new location (at least for a period). Notably, these are requirements that human cancer cells also must meet to metastasize. Recent studies show how DFTD evades the host immune system with a strategy something like getting past a doorman. DFTD suppresses antigen presentation—it doesn’t announce itself or sign in at the “front desk”—so the “doorman” doesn’t know that DFTD has entered. CTVT does something similar, except that it suppresses antigen presentation only at first, during the progressive phase; during the stabilization and regression phases, antigen presentation is largely restored.
Suppression of antigen presentation is a tactic that human cancer cells use to evade detection when they metastasize from a primary to a secondary tumor site. What we know about CTVT suggests that restoring antigen presentation could reestablish disease control. At present, a number of agents that can bolster antigen presentation are under investigation for the treatment of human cancer. Such an immunotherapeutic approach could work, but the jury is still out.
It has also been shown that telomere length in DFTD cells is regulated. In simple terms, telomeres are to chromosomes what aglets (the protective tips) are to shoelaces; telomeres protect chromosome ends from damage, such as degradation or aberrant fusions with other chromosomes. Telomere length correlates with replication potential: the longer the telomeres, the more times a cell can divide. Telomeres in DFTD cells aren’t longer than those in normal Tasmanian devil cells, but they can be elongated when they get too short. (Imagine replacing an aglet when it starts to wear out.) Furthermore, telomere length in DFTD cells appears to be stabilized by a protective protein complex. (Imagine also wrapping an aglet in duct tape after replacement.) In human cancers, aberrant telomere homeostasis is a common feature. Efforts are underway to target telomerase, the enzyme responsible for lengthening short telomeres, as one approach to fight cancer.
What are the implications of unusual animal cancers for human cancer biology?
The existence of clonally transmissible cancers means that cancer can be a disease of a species. One could argue that such a cancer arose in Tasmanian devils because they are highly inbred and genetically similar. But such a cancer also arose in and spread successfully among genetically diverse canine breeds, suggesting that susceptibility of a species to this type of cancer is not restricted to those that are inbred. At present, it’s not known how DFTD or CTVT originally arose, or what specific mutations conferred their capacity to thrive in this unusual way, so it’d be pure speculation whether this could happen in another animal species, including humans. But considering that the clonally transmissible cancers and human cancers share common escape and survival mechanisms, we speculate that it is possible, although highly unlikely (since this has only arisen twice so far in the known history of human and animal disease studies). We don’t fully understand yet how most dogs are able to keep this cancer in check, but that they do is evidence of the strength and plasticity of the immune system. By investigating the origins of these cancers, observing their evolution, and understanding their interactions with their respective hosts, we can gain a broader view of cancer biology and how we can fight it better in humans.
| The capacity of clonal cancer cells to be passed through a species attests to the awesome power of natural selection, which has led these cancers to evolve to be fit enough to survive for millennia. Sure, there are any number of organisms that have been around for at least that long, but the clonal cancers survive without sexual reproduction, an important mechanism used by many species to mix genes in new combinations. In some ways, we can liken these clonal cancers to an asexually reproducing organism, in which offspring are clones of the parent. |
The future of the devils hangs in the balance.
From an evolutionary standpoint, it doesn’t benefit the DFTD cancer to kill off its host too quickly or completely, because then it too would perish. Extrapolating from the CTVT experience, it’s conceivable that DFTD and the devil could co-evolve to balance tumor growth with suppression by the host immune system. Given enough time, this could happen… but the Tasmanian devils’ advocates aren’t waiting for nature to run its course; they’re already working on a vaccine. Let’s hope investigators make it in time to save the devils.
Here’s how you can help.
